Hui Zheng's team discovered a new protein post-translational modification-protein pyruvylation, and revealed the mechanism by which high glucose inhibits antiviral immunity
Source:Hui Zheng
2026-04-23
On February 27, 2026, a research paper titled "Pyruvate is a natural suppressor of interferon signaling by inducing STAT1 protein pyruvylation" was published in the journal Cell by the team led by Professor Hui Zheng from the School of Medicine at the University of Electronic Science and Technology of China, and the Sichuan Academy of Medical Sciences and Sichuan Provincial People's Hospital. The study identified a novel protein post-translational modification—protein pyruvylation, and thoroughly elucidated the molecular mechanism by which STAT1 pyruvylation inhibits type I interferon (IFN-I) antiviral immunity. This discovery reveals key mechanisms by which both antiviral immunity in individuals with hyperglycemia was reduced and individuals with hyperglycemia and hepatitis B infection are insensitive to IFN-I therapy, opening new directions in the field of metabolism-immunity intersection and clinical infection prevention and control.
Glycolysis is the core metabolic pathway of life. Glucose generates pyruvate through glycolysis. Its metabolic abnormalities are closely related to diabetes, cancer and other major diseases. However, it has not been reported whether pyruvate can modify substrate proteins to produce protein acetylation modification. In addition, clinical data show that hyperglycemia will aggravate severe viral infections such as influenza and COVID-19, but the underlying molecular mechanisms are still unclear.
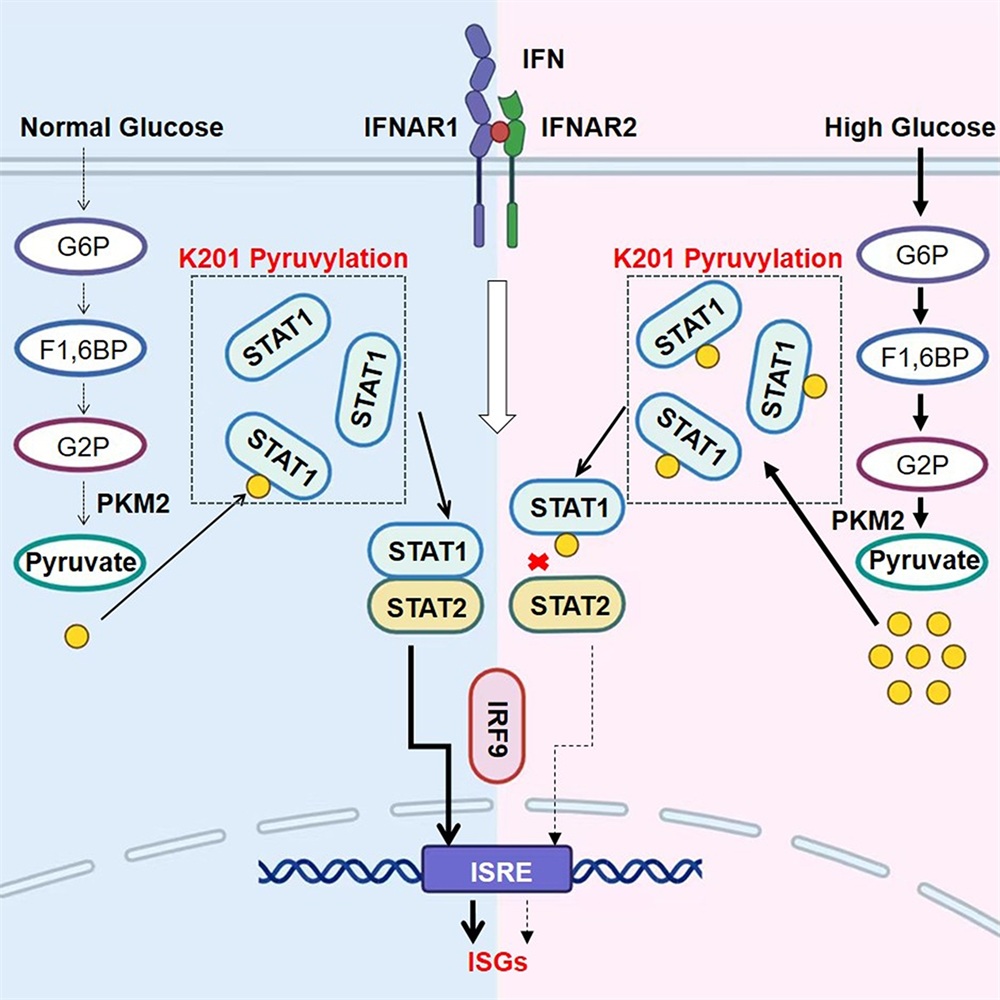
Hui Zheng's team has demonstrated for the first time that the glycolytic product pyruvate can covalently modify substrate proteins, forming protein pyruvylation modification, and thereby inhibiting the IFN-I signaling pathway and interferon-stimulated genes (ISGs) expression, weakening the body's antiviral defense. The team further identified STAT1 K201 as a key site for pyruvylation modification through mass spectrometry. Mechanistically, high glucose promotes the pyruvylation modification of STAT1, which hinders the binding of STAT1 to STAT2, thereby blocking IFN-I signaling and significantly inhibiting the body's IFN-I antiviral immunity. To analyze the in vivo function of this modification, the team conducted in vivo experiments using STAT1-K201R (KI) mice. The results showed that the pyruvylation level of STAT1 in various organs of KI mice was significantly reduced, and the expression of ISGs induced by IFN-I was higher; The virus infection model showed that KI mice had a stronger IFN-I immune response and ultimately lower viral load. Clinical sample analysis found that the pyruvylation modification of STAT1 in peripheral blood mononuclear cells (PBMCs) of patients with hyperglycemia was significantly increased, and the modification level was significantly negatively correlated with the expression of ISGs.
It is worth noting that protein pyruvylation modification is a widely present protein post-translational modification. Pyruvate is not only a core metabolic molecule, but also plays a role as an immune signaling regulatory factor. This discovery updates people's understanding of the function of pyruvate, linking metabolic regulation with innate immunity and providing a new paradigm for understanding physiological homeostasis and disease mechanisms. From a clinical value perspective, this research provides new ideas and intervention targets for enhancing antiviral immunity and preventing severe viral infections (such as viral sepsis) in individuals with high blood sugar; It has an important transformation value in optimizing the interferon treatment scheme for chronic viral infections such as hepatitis B, and improving the clinical prognosis of patients with abnormal blood sugar. In the future, in-depth analysis of protein pyruvylation modification is expected to lead to new diagnosis and treatment strategies in metabolic diseases, infectious diseases, tumors, autoimmune diseases and other fields.
Article link: https://www.cell.com/cell/fulltext/S0092-8674(26)00453-8
Glycolysis is the core metabolic pathway of life. Glucose generates pyruvate through glycolysis. Its metabolic abnormalities are closely related to diabetes, cancer and other major diseases. However, it has not been reported whether pyruvate can modify substrate proteins to produce protein acetylation modification. In addition, clinical data show that hyperglycemia will aggravate severe viral infections such as influenza and COVID-19, but the underlying molecular mechanisms are still unclear.
Hui Zheng's team has demonstrated for the first time that the glycolytic product pyruvate can covalently modify substrate proteins, forming protein pyruvylation modification, and thereby inhibiting the IFN-I signaling pathway and interferon-stimulated genes (ISGs) expression, weakening the body's antiviral defense. The team further identified STAT1 K201 as a key site for pyruvylation modification through mass spectrometry. Mechanistically, high glucose promotes the pyruvylation modification of STAT1, which hinders the binding of STAT1 to STAT2, thereby blocking IFN-I signaling and significantly inhibiting the body's IFN-I antiviral immunity. To analyze the in vivo function of this modification, the team conducted in vivo experiments using STAT1-K201R (KI) mice. The results showed that the pyruvylation level of STAT1 in various organs of KI mice was significantly reduced, and the expression of ISGs induced by IFN-I was higher; The virus infection model showed that KI mice had a stronger IFN-I immune response and ultimately lower viral load. Clinical sample analysis found that the pyruvylation modification of STAT1 in peripheral blood mononuclear cells (PBMCs) of patients with hyperglycemia was significantly increased, and the modification level was significantly negatively correlated with the expression of ISGs.

It is worth noting that protein pyruvylation modification is a widely present protein post-translational modification. Pyruvate is not only a core metabolic molecule, but also plays a role as an immune signaling regulatory factor. This discovery updates people's understanding of the function of pyruvate, linking metabolic regulation with innate immunity and providing a new paradigm for understanding physiological homeostasis and disease mechanisms. From a clinical value perspective, this research provides new ideas and intervention targets for enhancing antiviral immunity and preventing severe viral infections (such as viral sepsis) in individuals with high blood sugar; It has an important transformation value in optimizing the interferon treatment scheme for chronic viral infections such as hepatitis B, and improving the clinical prognosis of patients with abnormal blood sugar. In the future, in-depth analysis of protein pyruvylation modification is expected to lead to new diagnosis and treatment strategies in metabolic diseases, infectious diseases, tumors, autoimmune diseases and other fields.
Article link: https://www.cell.com/cell/fulltext/S0092-8674(26)00453-8


