Dr. Chunliang Xu’s team discovered fasting-induced plasma cell depletion impairs humoral immunological memory in Immunity
Source:Chunliang Xu
2026-04-21
On April 14, 2026, Dr. Chunliang Xu’s team at Zhongshan School of Medicine, Sun Yat-sen University published a research article in Immunity journal titled “Fasting impairs humoral immunological memory by β-hydroxybutyrate-mediated depletion of plasma cells.” The study reveals that fasting drive depletion of bone marrow plasma cells through the metabolic product β-hydroxybutyrate (BHB), thereby impairing humoral immunological memory. These findings provide new evidence linking metabolism to the durability of vaccine protection.
Humoral immunological memory underlies long-term vaccine protection and depends on long-lived plasma cells (LLPCs), which reside in bone marrow niche and continuously secrete protective antibodies. Although the strength and duration of vaccine protection vary substantially across vaccines and individuals, the mechanisms governing LLPC survival have remained largely unclear. Fasting, a popular dietary intervention, is often associated with potential benefits such as improved metabolism and delayed aging. However, whether it affects humoral immune memory had remained unknown.
To address this question, the research team established several fasting regimens in mice, including alternate-day fasting (ADF), 16/8 time-restricted feeding, and prolonged fasting. Across these models, fasting consistently caused a significant reduction in bone marrow plasma cells. In both vaccination and infection settings, intermittent fasting significantly reduced antigen-specific antibody titers and the number of antigen-specific LLPCs in the bone marrow, while also weakening vaccine-induced protection. By contrast, fasting did not alter the number of memory B cells or impair recall responses. These results indicate that fasting compromises humoral immunological memory primarily by promoting LLPC depletion.
Furthermore, the ketogenic diet could phenocopy even in the absence of fasting, leading to the decreased serum antigen-specific antibody levels and bone marrow LLPC number. Mechanistically, both fasting and ketogenic diet increased ketone body levels, with BHB emerging. The study demonstrated that BHB acts as a signaling metabolite and directly engages hydroxycarboxylic acid receptor 2 (HCAR2) on plasma cells to drive this effect.
To define how fasting induces bone marrow plasma cell depletion, the research team systematically examined plasma cell generation, homing, and maintenance. Fasting did not substantially alter plasma cell production or their homing to the bone marrow, nor did it directly induce plasma cell death within the bone marrow. Instead, RNA-seq analysis revealed that fasting mainly reduced expression of the key retention factor CXCR4 on plasma cells. This weakened LLPC retention within the protective bone marrow niche, promoted their mobilization into the circulation, and ultimately led to apoptosis. At the molecular level, BHB activated HCAR2 on plasma cells and suppressed cAMP signaling through the HCAR2-Gαi-AC-cAMP axis, thereby downregulating CXCR4. Pharmacologic activation of adenylate cyclase with forskolin restored CXCR4 expression and reversed fasting-induced plasma cell depletion and peripheral mobilization.
The research team also extended these observations beyond mouse model by examining the impact of fasting on human volunteers. Compared with controls, individuals undergoing structured fasting regimens displayed significantly accelerated declines in antibodies against multiple vaccine- or infection-related antigens. These observations suggest that the effect of fasting on human humoral immunological memory may parallel what was observed in mice.
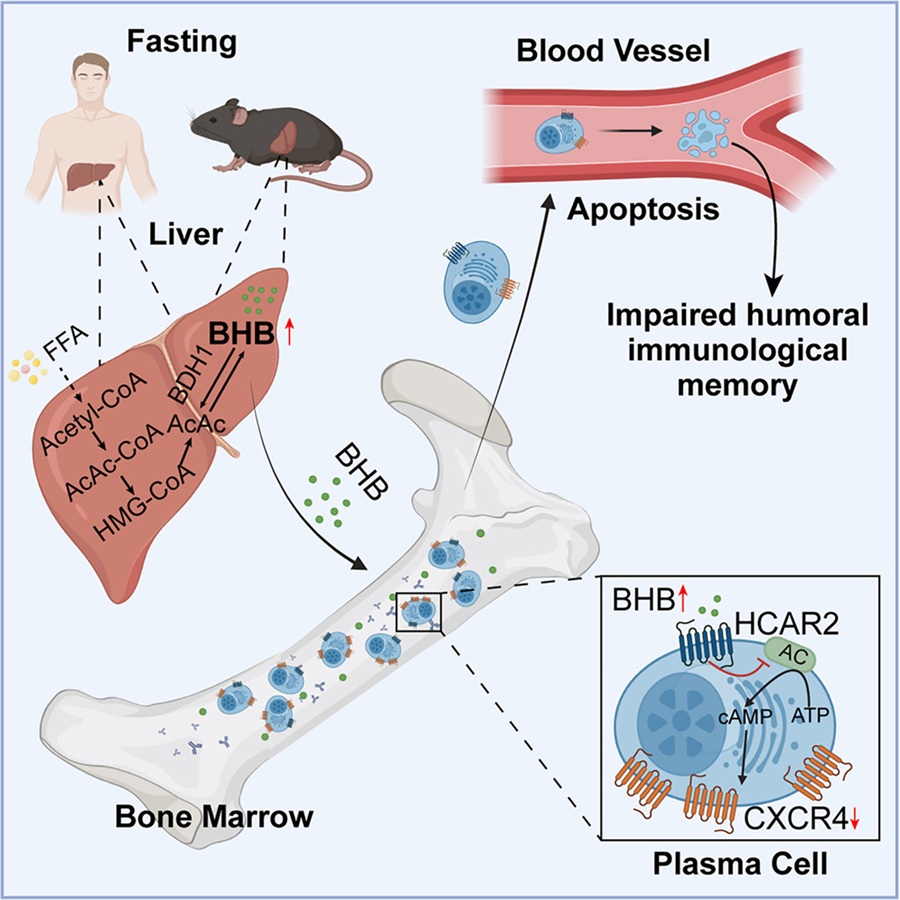
Taken together, this study systematically delineates a mechanism by which fasting induces BHB signaling, depletes bone marrow LLPCs, and impairs humoral immunological memory. This work highlights dietary patterns and lifestyle as potential determinants of the durability of vaccine protection, advances the understanding of the mechanisms that sustain long-lived plasma cells, and provides a new conceptual framework for future vaccine design and immunotherapeutic strategies.
Dr. Chunliang Xu from Zhongshan School of Medicine, Sun Yat-sen University, is the corresponding author of this paper, and doctoral candidate Yiming Zhu is the first author. The research process also received generous support from Professors Hui Zhang, Yiping Li, Yiwen Zhang, and Nan Cao at Sun Yat-sen University; Professor Hua Jin at Nanfang Hospital of Southern Medical University; Professor Jijun Zhao at the First Affiliated Hospital of Sun Yat-sen University; and Professor Hai Qi at Tsinghua University.
Article link: https://www.cell.com/immunity/fulltext/S1074-7613(26)00003-8
Humoral immunological memory underlies long-term vaccine protection and depends on long-lived plasma cells (LLPCs), which reside in bone marrow niche and continuously secrete protective antibodies. Although the strength and duration of vaccine protection vary substantially across vaccines and individuals, the mechanisms governing LLPC survival have remained largely unclear. Fasting, a popular dietary intervention, is often associated with potential benefits such as improved metabolism and delayed aging. However, whether it affects humoral immune memory had remained unknown.
To address this question, the research team established several fasting regimens in mice, including alternate-day fasting (ADF), 16/8 time-restricted feeding, and prolonged fasting. Across these models, fasting consistently caused a significant reduction in bone marrow plasma cells. In both vaccination and infection settings, intermittent fasting significantly reduced antigen-specific antibody titers and the number of antigen-specific LLPCs in the bone marrow, while also weakening vaccine-induced protection. By contrast, fasting did not alter the number of memory B cells or impair recall responses. These results indicate that fasting compromises humoral immunological memory primarily by promoting LLPC depletion.
Furthermore, the ketogenic diet could phenocopy even in the absence of fasting, leading to the decreased serum antigen-specific antibody levels and bone marrow LLPC number. Mechanistically, both fasting and ketogenic diet increased ketone body levels, with BHB emerging. The study demonstrated that BHB acts as a signaling metabolite and directly engages hydroxycarboxylic acid receptor 2 (HCAR2) on plasma cells to drive this effect.
To define how fasting induces bone marrow plasma cell depletion, the research team systematically examined plasma cell generation, homing, and maintenance. Fasting did not substantially alter plasma cell production or their homing to the bone marrow, nor did it directly induce plasma cell death within the bone marrow. Instead, RNA-seq analysis revealed that fasting mainly reduced expression of the key retention factor CXCR4 on plasma cells. This weakened LLPC retention within the protective bone marrow niche, promoted their mobilization into the circulation, and ultimately led to apoptosis. At the molecular level, BHB activated HCAR2 on plasma cells and suppressed cAMP signaling through the HCAR2-Gαi-AC-cAMP axis, thereby downregulating CXCR4. Pharmacologic activation of adenylate cyclase with forskolin restored CXCR4 expression and reversed fasting-induced plasma cell depletion and peripheral mobilization.
The research team also extended these observations beyond mouse model by examining the impact of fasting on human volunteers. Compared with controls, individuals undergoing structured fasting regimens displayed significantly accelerated declines in antibodies against multiple vaccine- or infection-related antigens. These observations suggest that the effect of fasting on human humoral immunological memory may parallel what was observed in mice.

Taken together, this study systematically delineates a mechanism by which fasting induces BHB signaling, depletes bone marrow LLPCs, and impairs humoral immunological memory. This work highlights dietary patterns and lifestyle as potential determinants of the durability of vaccine protection, advances the understanding of the mechanisms that sustain long-lived plasma cells, and provides a new conceptual framework for future vaccine design and immunotherapeutic strategies.
Dr. Chunliang Xu from Zhongshan School of Medicine, Sun Yat-sen University, is the corresponding author of this paper, and doctoral candidate Yiming Zhu is the first author. The research process also received generous support from Professors Hui Zhang, Yiping Li, Yiwen Zhang, and Nan Cao at Sun Yat-sen University; Professor Hua Jin at Nanfang Hospital of Southern Medical University; Professor Jijun Zhao at the First Affiliated Hospital of Sun Yat-sen University; and Professor Hai Qi at Tsinghua University.
Article link: https://www.cell.com/immunity/fulltext/S1074-7613(26)00003-8


