Sensory Neurons Drive Immune Exclusion in Triple-Negative Breast Cancer
Source:Yi-Zhou Jiang
2026-04-07
On February 6, 2026, a research team led by Prof. Yi-Zhou Jiang, Prof. Zhi-Ming Shao, and Associate Prof. Yi Xiao from Fudan University Shanghai Cancer Center, in collaboration with Prof. Jinfei Ni from the Institute for Translational Brain Research at Fudan University, published a study in Cell entitled “Sensory neurons drive immune exclusion by stimulating a dense extracellular matrix in the breast cancer tumor microenvironment.” The study demonstrates that tumor-infiltrating sensory neurons drive resistance to immunotherapy in triple-negative breast cancer (TNBC) and identifies a potential therapeutic strategy to overcome treatment resistance.
TNBC accounts for approximately 15–20% of breast cancers and is characterized by aggressive progression and a high risk of recurrence and metastasis. Although PD-1/PD-L1 checkpoint inhibitors have provided new therapeutic opportunities, a substantial proportion of patients still exhibit limited response or develop resistance. The mechanisms underlying this resistance remain incompletely understood.
In this study, analysis of 360 clinical TNBC samples revealed that perineural invasion (PNI) is strongly associated with poor prognosis and reduced response to immunotherapy. PNI-positive tumors displayed a typical immune-excluded phenotype, characterized by reduced immune cell infiltration and increased stromal fibrosis. Further analyses identified sensory neurons as the dominant neural subtype in these tumors, with their enrichment closely associated with resistance to anti–PD-1 therapy.
Mechanistic studies further showed that tumor-derived signals activate sensory neurons, which in turn stimulate cancer-associated fibroblasts to produce collagen and other extracellular matrix components. This process leads to the formation of a dense stromal barrier that prevents immune cells from entering the tumor core and thereby drives immune exclusion.
Consistently, inhibition of sensory neuron signaling in animal models reduced stromal fibrosis, enhanced immune cell infiltration, and suppressed tumor growth. Importantly, combining sensory neuron inhibition with immunotherapy significantly improved antitumor efficacy.
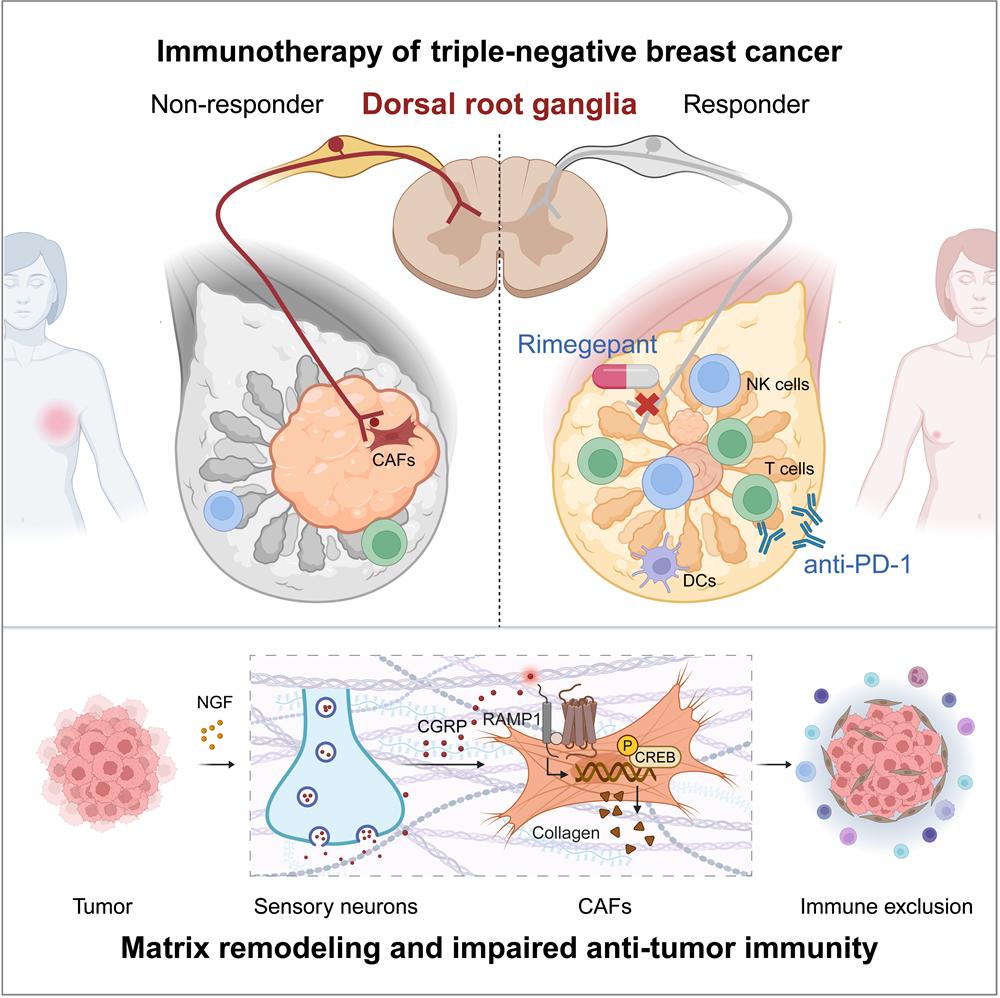
The study also highlights the translational potential of targeting this pathway. The CGRP receptor antagonist rimegepant, an approved drug for migraine treatment, may represent a promising candidate for drug repurposing in cancer therapy.
Taken together, this work identifies a sensory neuron–mediated stromal remodeling mechanism underlying immune exclusion in TNBC and provides a new perspective for improving immunotherapy responses.
Prof. Yi-Zhou Jiang, Prof. Zhi-Ming Shao, Prof. Jinfei Ni, and Associate Prof. Yi Xiao are the co-corresponding authors. Dr. Siwei Zhang, Dr. Han Wang, Associate Prof. Yi Xiao, and Dr. Luotian Liu are the co-first authors. This work was supported by the National Natural Science Foundation of China and the Fundamental and Interdisciplinary Disciplines Breakthrough Plan of the Ministry of Education of China, among other funding sources.
Original article: https://www.cell.com/cell/fulltext/S0092-8674(26)00048-6
TNBC accounts for approximately 15–20% of breast cancers and is characterized by aggressive progression and a high risk of recurrence and metastasis. Although PD-1/PD-L1 checkpoint inhibitors have provided new therapeutic opportunities, a substantial proportion of patients still exhibit limited response or develop resistance. The mechanisms underlying this resistance remain incompletely understood.
In this study, analysis of 360 clinical TNBC samples revealed that perineural invasion (PNI) is strongly associated with poor prognosis and reduced response to immunotherapy. PNI-positive tumors displayed a typical immune-excluded phenotype, characterized by reduced immune cell infiltration and increased stromal fibrosis. Further analyses identified sensory neurons as the dominant neural subtype in these tumors, with their enrichment closely associated with resistance to anti–PD-1 therapy.
Mechanistic studies further showed that tumor-derived signals activate sensory neurons, which in turn stimulate cancer-associated fibroblasts to produce collagen and other extracellular matrix components. This process leads to the formation of a dense stromal barrier that prevents immune cells from entering the tumor core and thereby drives immune exclusion.
Consistently, inhibition of sensory neuron signaling in animal models reduced stromal fibrosis, enhanced immune cell infiltration, and suppressed tumor growth. Importantly, combining sensory neuron inhibition with immunotherapy significantly improved antitumor efficacy.

The study also highlights the translational potential of targeting this pathway. The CGRP receptor antagonist rimegepant, an approved drug for migraine treatment, may represent a promising candidate for drug repurposing in cancer therapy.
Taken together, this work identifies a sensory neuron–mediated stromal remodeling mechanism underlying immune exclusion in TNBC and provides a new perspective for improving immunotherapy responses.
Prof. Yi-Zhou Jiang, Prof. Zhi-Ming Shao, Prof. Jinfei Ni, and Associate Prof. Yi Xiao are the co-corresponding authors. Dr. Siwei Zhang, Dr. Han Wang, Associate Prof. Yi Xiao, and Dr. Luotian Liu are the co-first authors. This work was supported by the National Natural Science Foundation of China and the Fundamental and Interdisciplinary Disciplines Breakthrough Plan of the Ministry of Education of China, among other funding sources.
Original article: https://www.cell.com/cell/fulltext/S0092-8674(26)00048-6


