The Wang Lab Reveals the Neuroimmune Mechanism of the “Inflammation-Anxiety” Cycle in Atopic Dermatitis in Immunity
Source:Fang Wang
2026-03-18
On February 20, 2026, a collaborative study led by Dr. Fang Wang (Dermatology Hospital of Southern Medical University), Dr. Fengxian Li (Zhujiang Hospital of Southern Medical University), and the First Affiliated Hospital of Sun Yat-sen University was published in Immunity. The paper, titled “Hyperactivation of sympathetic nerves fuels basophil infiltration in atopic dermatitis,” systematically reveals how dermatitis-induced anxiety amplifies skin inflammation through a “sympathetic nerve-basophil axis.” This research opens new therapeutic avenues for patients with severe atopic dermatitis (AD) suffering from comorbid emotional disorders.
This work originated from the clinical observation that AD patients are often trapped in a vicious cycle of “anxiety/insomnia and exacerbation of skin lesion.” To explore the underlying biological mechanisms, the research team first established an MC903-induced AD mouse model and confirmed that as skin lesions worsened, the mice exhibited significant anxiety-like behaviors. However, the study found that this stress-induced exacerbation of dermatitis does not rely on the classical hypothalamic-pituitary-adrenal (HPA) axis; neither bilateral adrenalectomy nor pharmacological blockade of HPA axis signaling could alleviate the anxiety-driven exacerbation of skin inflammation. Utilizing retrograde tracing techniques with viruses and chemical tracers, the research team focused on the autonomic nervous system. They discovered that the sympathetic nerves innervating the ear skin (primarily originating from the superior cervical ganglion, SCG) were in a state of hyperactivation during the course of AD, with a significant increase in the density of sympathetic nerve fibers in the lesional area and a markedly elevated local concentration of the neurotransmitter norepinephrine (NE).
In terms of mechanistic exploration, flow cytometry confirmed that basophils are the key cells receiving the pro-inflammatory signals from sympathetic nerves. NE released by sympathetic nerves acts directly on the highly expressed β2-adrenergic receptors (ADRβ2) on basophils, acting like an “acceleration engine” to greatly enhance their motility and migration capacity within the skin. More critically, the activated basophils synthesize and secrete large amounts of the chemokine CCL6, which binds to its own receptor CCR1 to form a powerful positive feedback loop. This continuously recruits more basophils to the lesional site, leading to persistent and exacerbated inflammation. Conditional knockout of the Adrb2 gene in basophils or blockade of the CCL6/CCR1 chemotactic pathway in mice effectively severed this “neuro-immune” crosstalk, resulting in significant alleviation of skin inflammation.
Regarding therapeutic efficacy and clinical translation, this mechanism was highly consistently validated in human clinical samples. The research team conducted an in-depth analysis of AD patient samples and found a significantly increased density of sympathetic nerve fibers in the skin lesions, which were spatially in close proximity to basophils. The expression levels of ADRβ2, CCL23 (the human homolog of CCL6), and CCR1 in the peripheral blood basophils of patients were significantly and abnormally elevated, and they showed a significant positive correlation with the patients' anxiety (GAD-7), depression (PHQ-9), and sleep disorder (PSQI) scores, as well as the severity of skin lesions (EASI). This not only provides direct biological evidence for the concurrent treatment of psychological and physical symptoms but also suggests that lowering sympathetic nerve tension through psychological interventions such as digital cognitive behavioral therapy (dCBT), or developing targeted drugs against the sympathetic nerve-basophil axis in the future, will become crucial breakthroughs for disrupting the vicious “inflammation-anxiety” cycle in AD patients.
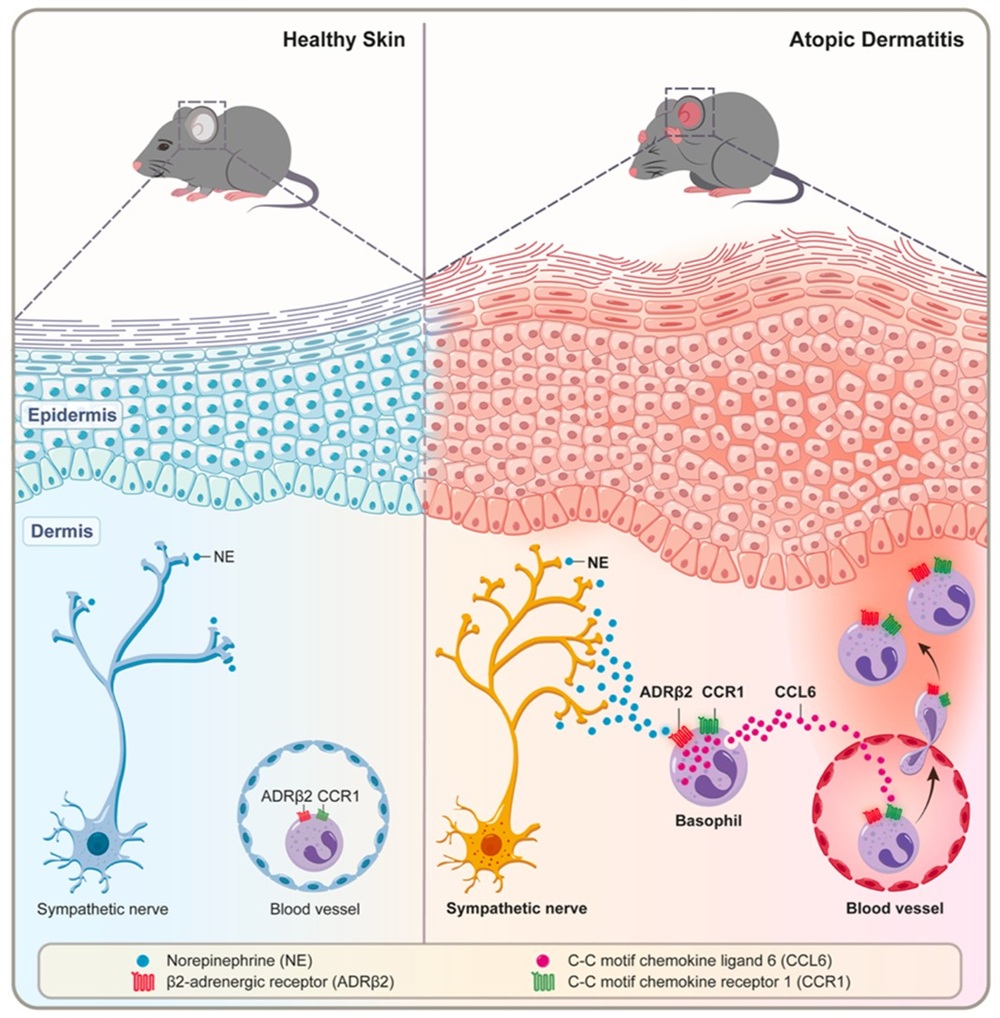
In summary, this study systematically elucidates the critical bridging role of the “sympathetic nerve-basophil axis” between emotion and skin inflammation, providing a new theoretical basis for the comprehensive treatment strategies of chronic inflammatory and pruritic skin diseases, and supplementing key evidence for the “brain-skin axis” interaction mechanism.
Dr. Fang Wang from the Dermatology Hospital of Southern Medical University and Dr. Fengxian Li from Zhujiang Hospital are the co-corresponding authors of this paper, and doctoral candidate Xinyang Xie is the first author. The research process also received experimental animal support from Professor Brian Kim at Mount Sinai Hospital in New York, Professor Wenwen Zeng and Professor Coco Chu at Tsinghua University, Professor Xia Dou at Peking University Shenzhen Hospital, Professor Liang Wang at Zhejiang University, Dr. Delu Che at Xi'an Jiaotong University, and Professor Xingjun Liu at Nantong University.
Article link: https://www.sciencedirect.com/science/article/pii/S1074761326000312
This work originated from the clinical observation that AD patients are often trapped in a vicious cycle of “anxiety/insomnia and exacerbation of skin lesion.” To explore the underlying biological mechanisms, the research team first established an MC903-induced AD mouse model and confirmed that as skin lesions worsened, the mice exhibited significant anxiety-like behaviors. However, the study found that this stress-induced exacerbation of dermatitis does not rely on the classical hypothalamic-pituitary-adrenal (HPA) axis; neither bilateral adrenalectomy nor pharmacological blockade of HPA axis signaling could alleviate the anxiety-driven exacerbation of skin inflammation. Utilizing retrograde tracing techniques with viruses and chemical tracers, the research team focused on the autonomic nervous system. They discovered that the sympathetic nerves innervating the ear skin (primarily originating from the superior cervical ganglion, SCG) were in a state of hyperactivation during the course of AD, with a significant increase in the density of sympathetic nerve fibers in the lesional area and a markedly elevated local concentration of the neurotransmitter norepinephrine (NE).
In terms of mechanistic exploration, flow cytometry confirmed that basophils are the key cells receiving the pro-inflammatory signals from sympathetic nerves. NE released by sympathetic nerves acts directly on the highly expressed β2-adrenergic receptors (ADRβ2) on basophils, acting like an “acceleration engine” to greatly enhance their motility and migration capacity within the skin. More critically, the activated basophils synthesize and secrete large amounts of the chemokine CCL6, which binds to its own receptor CCR1 to form a powerful positive feedback loop. This continuously recruits more basophils to the lesional site, leading to persistent and exacerbated inflammation. Conditional knockout of the Adrb2 gene in basophils or blockade of the CCL6/CCR1 chemotactic pathway in mice effectively severed this “neuro-immune” crosstalk, resulting in significant alleviation of skin inflammation.
Regarding therapeutic efficacy and clinical translation, this mechanism was highly consistently validated in human clinical samples. The research team conducted an in-depth analysis of AD patient samples and found a significantly increased density of sympathetic nerve fibers in the skin lesions, which were spatially in close proximity to basophils. The expression levels of ADRβ2, CCL23 (the human homolog of CCL6), and CCR1 in the peripheral blood basophils of patients were significantly and abnormally elevated, and they showed a significant positive correlation with the patients' anxiety (GAD-7), depression (PHQ-9), and sleep disorder (PSQI) scores, as well as the severity of skin lesions (EASI). This not only provides direct biological evidence for the concurrent treatment of psychological and physical symptoms but also suggests that lowering sympathetic nerve tension through psychological interventions such as digital cognitive behavioral therapy (dCBT), or developing targeted drugs against the sympathetic nerve-basophil axis in the future, will become crucial breakthroughs for disrupting the vicious “inflammation-anxiety” cycle in AD patients.

In summary, this study systematically elucidates the critical bridging role of the “sympathetic nerve-basophil axis” between emotion and skin inflammation, providing a new theoretical basis for the comprehensive treatment strategies of chronic inflammatory and pruritic skin diseases, and supplementing key evidence for the “brain-skin axis” interaction mechanism.
Dr. Fang Wang from the Dermatology Hospital of Southern Medical University and Dr. Fengxian Li from Zhujiang Hospital are the co-corresponding authors of this paper, and doctoral candidate Xinyang Xie is the first author. The research process also received experimental animal support from Professor Brian Kim at Mount Sinai Hospital in New York, Professor Wenwen Zeng and Professor Coco Chu at Tsinghua University, Professor Xia Dou at Peking University Shenzhen Hospital, Professor Liang Wang at Zhejiang University, Dr. Delu Che at Xi'an Jiaotong University, and Professor Xingjun Liu at Nantong University.
Article link: https://www.sciencedirect.com/science/article/pii/S1074761326000312


